The year 2025 has seen a significant resurgence of Highly Pathogenic Avian Influenza (HPAI), commonly known as bird flu, prompting global concern. This outbreak, marked by its rapid spread and increased reports of mammalian infections, underscores the virus’s unpredictable nature and the urgent need for comprehensive understanding and control. Very specifically, the bird flu recent outbreak in the North American region causing severe impact on poultry and dairy industry with a 70 human cases in USA (as of March 2025).
Avian influenza, caused by influenza A viruses, naturally circulates in wild aquatic birds. While many strains are low pathogenic, causing mild or no symptoms, a subset can evolve into highly pathogenic forms capable of devastating poultry and posing a risk to other animals and humans. In this article, we will discuss an in-depth analysis of HPAI’s epidemiology, transmission dynamics, spatial and temporal distributions, and the ongoing challenges in its prevention and control.
What is Avian Influenza (Bird Flu)?
Avian influenza, commonly known as bird flu, refers to infections caused by avian influenza viruses (AIVs), which are highly contagious and exhibit significant genetic variability. These viruses predominantly circulate among avian populations, with wild aquatic birds serving as their primary natural reservoirs. Most bird flu viruses are classified as low pathogenic avian influenza (LPAI), typically resulting in mild or asymptomatic infections in poultry. However, a subset of these viruses can mutate into highly pathogenic avian influenza (HPAI) strains, which are characterized by extreme virulence, leading to mortality rates of 90–100% in poultry flocks, rapid epidemic outbreaks, and severe economic consequences due to trade restrictions.
While bird flu primarily affects birds, sporadic transmission to mammals, including humans, has been documented. Although the majority of human infections are mild or asymptomatic, certain viral lineages—such as H5N1 HPAI, which emerged in Asia, and H7N9 LPAI, identified in China—have raised significant public health concerns due to their potential for severe disease. Human infections typically result from direct, unprotected exposure to infected birds or contaminated environments, with clinical manifestations ranging from subclinical infection to fatal illness.
Epidemiology and Transmission
HPAI belongs to the Influenza A virus, categorized under the Orthomyxoviridae family, and is classified based on its hemagglutinin (HA) and neuraminidase (NA) proteins. While many avian influenza viruses (AIVs) exhibit low pathogenicity, certain strains, such as H5N1, have evolved into highly virulent forms through mutations and genetic reassortments.
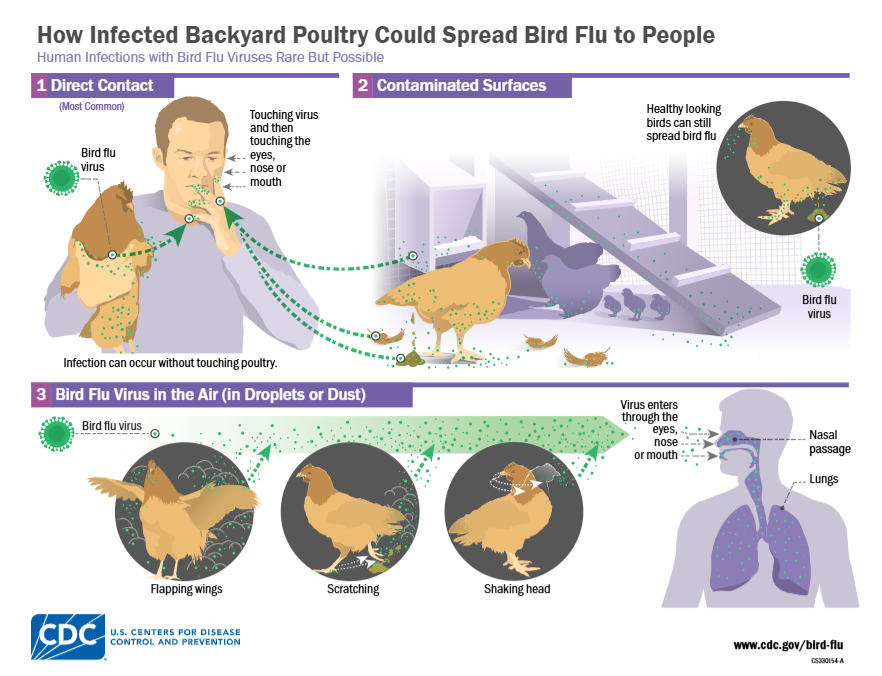
Hosts and Transmission Routes
- Primary Reservoirs: Wild birds, particularly those in the Anseriformes (ducks, geese, and swans) and Charadriiformes (shorebirds, gulls, and terns) orders, serve as natural hosts of bird flu.
- Poultry Impact: HPAI predominantly affects domestic poultry, such as chickens and turkeys, leading to 90–100% mortality rates, rapid flock devastation, and significant economic losses.
- Mammalian Transmission: Although primarily an avian disease, HPAI has been sporadically detected in mammals, including cats, dogs, foxes, minks, and cattle, raising concerns about cross-species transmission.
- Environmental Factors: High humidity and low temperatures facilitate viral persistence and transmission, allowing outbreaks to surge during cold seasons.
Modes of Transmission
HPAI spreads through multiple transmission routes, making containment a significant challenge:
- Direct Contact: Infection occurs when healthy birds come into physical contact with infected birds or their secretions, such as saliva, nasal discharge, and feces.
- Indirect Contact: Contaminated feed, water, surfaces, and fomites (e.g., equipment, clothing, and vehicles) serve as vectors for viral transmission.
- Airborne Transmission: Infected birds release viral particles that can become airborne and spread via inhalation of aerosols or contaminated droplets.
- Consumption of Raw Products: There is a known risk of transmission from consuming raw or undercooked products, including raw milk and raw milk derivatives, which have been linked to infections in mammals such as domestic cats.

Spatial Distribution and Global Impact
HPAI outbreaks have been recorded in over 60 countries, severely affecting poultry industries and leading to trade restrictions and economic losses. In Asia, HPAI is endemic in several countries, including Bangladesh, China, and Vietnam, where the virus has evolved into diverse clades, with seasonal outbreaks linked to migratory bird pathways. In Europe and North America, migratory wild birds have introduced HPAI strains into previously unaffected regions, leading to outbreaks in both wild and domestic bird populations.
Although less prevalent in Africa, sporadic outbreaks have been reported, often associated with trade and migratory birds. The current HPAI outbreak in North America began in November 2021 when the virus was introduced from Europe—either through trans-Atlantic bird migration or viral transmission via Iceland and Greenland. Once introduced, the virus spread rapidly across wild and domestic bird populations, causing significant losses in the poultry industry.
Temporal Trends and Seasonal Outbreaks
HPAI exhibits distinct seasonal patterns, with outbreaks peaking during colder months when environmental conditions favor viral survival. Studies indicate that the virus persists longer in cold, dry conditions, with an optimal temperature range of 15–20°C for its spread. This has been observed in Bangladesh, where outbreaks predominantly occur between January and April, aligning with winter temperatures and increased human-poultry interactions in live bird markets. Similarly, across Southeast Asia, HPAI activity follows a seasonal cycle, often surging in the late autumn to early spring, coinciding with migratory bird movements that introduce new viral strains into domestic poultry populations.
In Europe and North America, outbreaks have demonstrated a cyclical pattern, typically escalating during colder months as migratory birds from infected regions arrive. The November 2021 outbreak in North America is a notable example, where the virus, introduced from Europe via migratory pathways, quickly spread through wild bird populations and subsequently infiltrated commercial poultry operations. In these regions, the virus is often detected in autumn and early winter, with cases declining as temperatures rise in late spring.
The role of wild bird migration is critical in the seasonal distribution of HPAI. Certain migratory flyways, such as the East Asian-Australasian Flyway and the Atlantic Flyway, serve as conduits for the long-distance dissemination of avian influenza viruses. Surveillance data reveal that waterfowl and shorebirds play a substantial role in carrying and introducing new virus strains, often coinciding with major seasonal migrations. Consequently, disease surveillance and biosecurity measures must be intensified ahead of peak migration periods to mitigate cross-border transmission risks.
Furthermore, meteorological factors such as humidity, precipitation, and wind patterns influence the dispersal and survival of the virus. Studies indicate that outbreaks are more frequent in dry, cold climates, whereas excessive humidity and rainfall can reduce viral stability. However, sporadic summer outbreaks have also been recorded, particularly in regions where biosecurity lapses allow the virus to persist within poultry farming systems.
Given these temporal trends, predictive modeling has become an essential tool in forecasting outbreak patterns and informing preventative strategies. By analyzing historical outbreak data, climatic conditions, and bird migration routes, researchers can identify high-risk periods and regions, allowing for preemptive vaccination campaigns, enhanced monitoring, and timely intervention measures.
Public Health Concerns and Zoonotic Potential
The risk of H5N1 avian influenza to human health remains a growing concern as sporadic human infections continue to be documented. The recent death of a patient in Louisiana marks the first known fatality in the U.S., raising alarms among public health experts. Genetic sequencing of the virus from this case revealed mutations that indicate adaptation toward human infection, a development that parallels previous cases in British Columbia and other regions. While human-to-human transmission has not yet been observed, these genetic changes heighten concerns about the virus’s potential to evolve into a form capable of sustained human transmission.
As of 2024, 66 confirmed human cases of H5N1 have been reported in the U.S., with most infections occurring among individuals with direct exposure to infected poultry or dairy farms. Although most cases have been mild, the increasing prevalence of H5N1 in mammals, including domestic cats and dairy cattle, signals an expanding host range that could facilitate further mutations. The potential for reassortment—where H5N1 exchanges genetic material with seasonal influenza viruses—poses an additional pandemic risk, as seen in previous global flu pandemics.
The presence of H5N1 in dairy cattle remains a particularly troubling development, with over 300 affected herds nationwide. Although efforts to control the outbreak have seen some success in regions like California, the virus’s continued spread through contaminated milking equipment and indirect transmission pathways necessitates enhanced biosecurity measures. The role of wild mammals, such as ferrets and field mice, in facilitating cross-species transmission also warrants further investigation.
While the risk to the general public remains low, individuals working in poultry farms, dairy operations, and veterinary settings face a higher risk of exposure. The need for enhanced surveillance, particularly through bulk milk tank testing and expanded testing of mammalian hosts, is crucial for early detection and containment. Public health agencies emphasize the importance of protective measures, such as mask-wearing, proper hygiene, and avoiding the consumption of raw milk or unprocessed dairy products, to mitigate individual risk.
Continued genetic monitoring of the virus is essential to detect changes that may signal increased transmissibility. If sustained human-to-human transmission were to occur, it would represent a significant global public health emergency, necessitating widespread containment efforts. Until then, proactive surveillance and strict biosecurity protocols remain our best defense against a potential H5N1 pandemic.
Control and Prevention Strategies
Eradication of HPAI remains challenging due to its ability to persist in wild bird populations and environmental reservoirs. The following strategy is reccomended at dififenent level of control for bird flu
Personal Level
- Consume only pasteurized dairy products to avoid potential exposure to the virus through contaminated milk.
- Properly cook eggs, meat, and poultry to recommended temperatures to prevent infection.
- Get vaccinated against seasonal influenza, reducing the risk of co-infection and reassortment with H5N1.
- Practice good hygiene, including frequent handwashing, especially after contact with birds, poultry, or outdoor areas where birds may gather.
- Avoid contact with sick or dead birds and report any findings to local wildlife or health authorities.
Farm Level
- Enhance biosecurity measures, such as restricting access to poultry farms, disinfecting equipment, and implementing strict sanitation protocols.
- Monitor and test poultry regularly for early detection of HPAI cases to prevent large-scale outbreaks.
- Separate domestic poultry from wild birds to minimize transmission risks.
- Dispose of infected birds properly and follow culling protocols recommended by veterinary health authorities.
- Provide protective gear (masks, gloves, boots) for farm workers handling poultry and ensure they follow safety guidelines.
Workplace Level (Poultry and Dairy Farms, Veterinary Clinics, and Livestock Facilities)
- Implement mandatory protective measures, including masks, gloves, and personal protective equipment (PPE) for workers handling animals.
- Conduct routine surveillance and testing for early detection of HPAI among farm and veterinary staff.
- Encourage flu vaccination among high-risk workers to reduce potential human-to-human transmission risks.
- Limit movement of personnel between farms to prevent cross-contamination.
- Train employees on proper biosecurity measures, ensuring they understand the risks and protocols to mitigate infection.
Government Level
- Strengthen surveillance programs to track the spread of HPAI in poultry, livestock, and wild bird populations.
- Regulate and enforce strict biosecurity measures in poultry and dairy industries.
- Develop and distribute vaccines targeting H5N1 for high-risk populations, including farm workers and veterinarians.
- Increase public awareness campaigns to educate individuals on preventive measures and the risks associated with HPAI.
- Coordinate international efforts to monitor HPAI outbreaks and implement joint containment strategies.
Challenges and Future Directions
The role of migratory birds in global HPAI dissemination necessitates international collaboration for surveillance and control. The use of antiviral drugs in poultry has raised concerns about the emergence of resistant strains. One Health Approach by integrating animal and human health surveillance systems is essential for early warning and response to potential zoonotic spillovers. A One Health approach that bridges veterinary, environmental, and human health sectors is crucial for mitigating the spread of HPAI. By fostering interdisciplinary collaboration and global coordination, we can strengthen outbreak preparedness, enhance biosecurity measures, and develop targeted interventions to prevent future pandemics.
Conclusion
HPAI remains a significant global threat with profound implications for poultry industries, public health, and biodiversity. Strengthening biosecurity, enhancing surveillance, and fostering international cooperation are crucial steps toward mitigating its impact. While eradication remains challenging, a comprehensive approach integrating epidemiology, virology, and policy measures can aid in controlling HPAI and reducing its zoonotic risks
Learn more on Bird Flu at CDC website