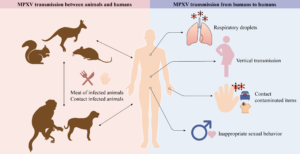
Did you know that over 60% of human infectious diseases originate in animals? This statistic highlights a reality we encounter daily, from the pets we cuddle to the food we eat. Today, we will be discussing zoonotic diseases—diseases that can spread from animals to humans. Before jumping into the main discussion, let’s clarify some key terms:
- Zoonoses: According to the WHO, a disease that can spread from animals to humans.
- Zoonotic Pathogens: The pathogen/causal agent responsible for zoonoses.
- Reservoir: An animal host that carries a pathogen without displaying significant symptoms, serving as a source of infection for other hosts.
- Spillover: The transmission of a pathogen from a reservoir host to a new, susceptible host species.
What are zoonotic diseases?
Simply put, they are diseases or infections that can be transmitted from animals to humans. These can be caused by various pathogens, including viruses, bacteria, parasites, fungi, and even prions. Now that we’ve defined zoonoses and clarified the terminology, let’s explore how these diseases are transmitted. Zoonotic diseases can spread from animals to humans through several routes:
- Direct Transmission: This occurs through direct contact with an infected animal’s bodily fluids, such as blood, saliva, or urine, or through bites or scratches. Examples include: Rabies (through bites), Brucellosis (through contact with infected animal fluids)
- Indirect Transmission: This involves contact with contaminated surfaces or objects (fomites) in the environment where infected animals have been. Examples include: Leptospirosis (through contaminated water or soil) and Anthrax (through contaminated soil or animal products)
- Vector-Borne Transmission: This occurs when an infected vector, such as a mosquito, tick, or flea, transmits a pathogen from an animal to a human. Examples include: Lyme disease (ticks), West Nile virus (mosquitoes), Plague (fleas)
- Foodborne Transmission: This involves consuming food contaminated with a zoonotic pathogen. Examples include: Salmonellosis (contaminated meat, eggs), E. coli infections (contaminated food or water), Campylobacteriosis (contaminated poultry)
- Waterborne Transmission: This occurs through the ingestion of water contaminated with a zoonotic pathogen. Examples include: Giardiasis (contaminated water), Cryptosporidiosis (contaminated water)
- Environmentally Transmitted: This involves exposure to pathogens present in the environment due to animal contamination. Examples include: Histoplasmosis (fungal spores in soil contaminated with bird or bat droppings), Ascariasis (parasitic eggs in contaminated soil)
Global Prioritization of Zoonotic Diseases
The global burden of zoonotic diseases necessitates a systematic approach to prioritization and intervention. To this end, numerous countries have implemented the Centers for Disease Control and Prevention’s (CDC) One Health Zoonotic Disease Prioritization (OHZDP) process. This multidisciplinary framework facilitates the identification of high-priority zoonotic pathogens within specific national contexts. As of February 2025, a total of 42 countries have published their OHZDP findings on the CDC website, providing a valuable dataset for comparative analysis.
Despite variations in geographical and ecological factors, a significant degree of overlap is observed in the zoonotic diseases most frequently prioritized across these nations. Diseases includes:
Rabies: A Prevalent Zoonotic Threat
Rabies has emerged as a near-universal priority pathogen across diverse national settings. Its persistent presence in animal reservoir hosts, coupled with its associated high case-fatality rate in humans, contributes to its prominence in prioritization exercises. Although rabies exhibits a low pandemic potential, it is estimated to cause approximately 60,000 human deaths annually. In developing countries, the primary mode of transmission is through canine bites, whereas in developed countries, wildlife rabies, involving reservoir hosts such as bats, foxes, and raccoons, represents a significant public health concern.
The global health community, with the World Health Organization (WHO) playing a leading role, has established a target of eliminating dog-mediated human rabies by 2030. However, the achievement of this objective is contingent upon addressing several key challenges, including:
- Limited access to post-exposure prophylaxis (PEP) in resource-constrained environments.
- Difficulties in attaining comprehensive canine vaccination coverage.
- Underreporting of animal bite incidents and rabies cases.
- The complexities inherent in wildlife rabies management.
Continued research and collaborative initiatives are essential to overcome the challenges of rabies control towards the 2030 elimination target.
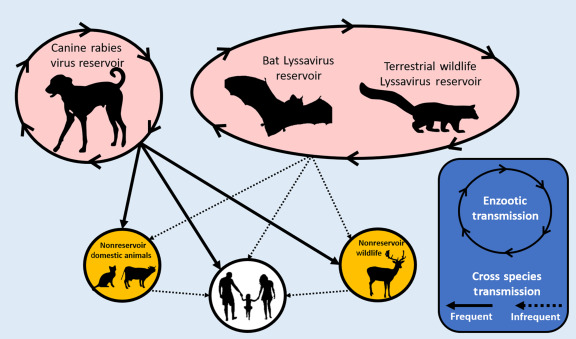
Figure: Tranmission of rabies in different host (Source: ScienceDirect)
Zoonotic Influenza Viruses: A Persistent Global Health Concern
Zoonotic influenza viruses remain a significant public health priority worldwide due to their inherent ability to cross species barriers. The recent impact of avian influenza on poultry industries, particularly in the United States, has underscored the potential economic and public health consequences of these viruses. Primarily circulating among avian and swine populations, these viruses can occasionally infect humans, leading to a spectrum of illnesses, including severe respiratory disease and, in some cases, pandemics.
A prominent example is the highly pathogenic avian influenza (HPAI) H5N1 strain, which poses a substantial risk due to its potential for zoonotic transmission from infected poultry to humans. While sustained human-to-human transmission of H5N1 remains limited, the virus’s propensity for mutation and adaptation necessitates ongoing vigilance. Similarly, swine influenza viruses, such as the one responsible for the 2009 H1N1 pandemic, represent a continuous threat.
Effective control strategies involve robust surveillance in animal populations, implementation of stringent biosecurity measures on farms, development and deployment of effective vaccines, and rapid response to human cases. A ‘One Health’ approach, as emphasized in Rahman et al. (2020), is crucial for addressing the complex interplay between animal, human, and environmental health in the context of zoonotic influenza
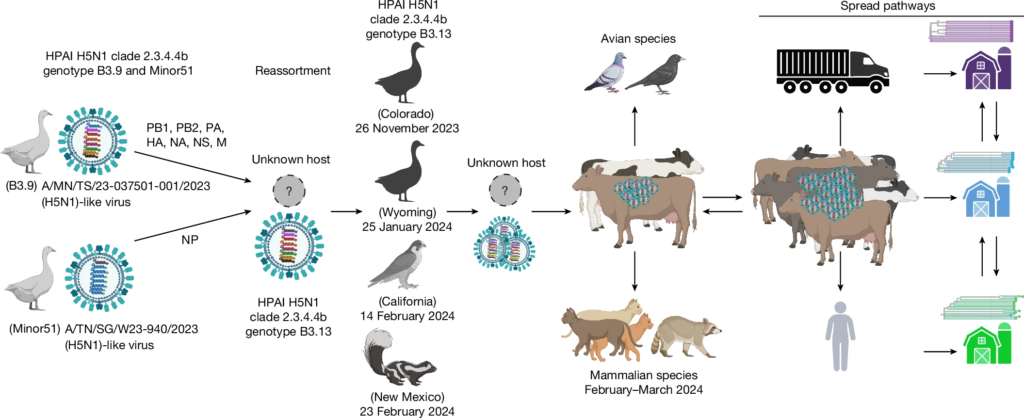
This model, proposed in a recent Nature publication, depicts the potential transmission of avian influenza viruses in dairy cattle in the United States, highlighting the need for continued research into zoonotic influenza transmission dynamics.
Brucellosis: A Persistent Zoonotic Threat
Brucellosis, a bacterial infection primarily affecting livestock, is a significant zoonotic disease prioritized globally due to its substantial economic impact and human health burden. The disease is caused by various Brucella species, which can infect a wide range of animals, including cattle, sheep, goats, and pigs. Humans typically contract brucellosis through the consumption of unpasteurized dairy products or close contact with infected animals.
The impact of brucellosis is multifaceted:
- Economic Impact: Brucellosis causes significant economic losses in the livestock industry due to reduced milk production, infertility, and abortions.
- Human Health Burden: Human brucellosis can manifest as a debilitating chronic illness with a wide range of symptoms, including fever, fatigue, joint pain, and depression.
- Global Distribution: Brucellosis is endemic in many parts of the world, particularly in developing countries with limited veterinary and public health infrastructure.
Controlling brucellosis requires a comprehensive approach involving:
- Vaccination of Livestock: Vaccination programs are essential for preventing brucellosis in animal populations.
- Surveillance and Testing: Regular surveillance and testing of livestock herds are crucial for early detection and control of outbreaks.
- Pasteurization of Dairy Products: Pasteurization effectively eliminates Brucella bacteria from milk and dairy products.
- Public Awareness: Educating the public about the risks of brucellosis and preventive measures is essential for reducing human infections.
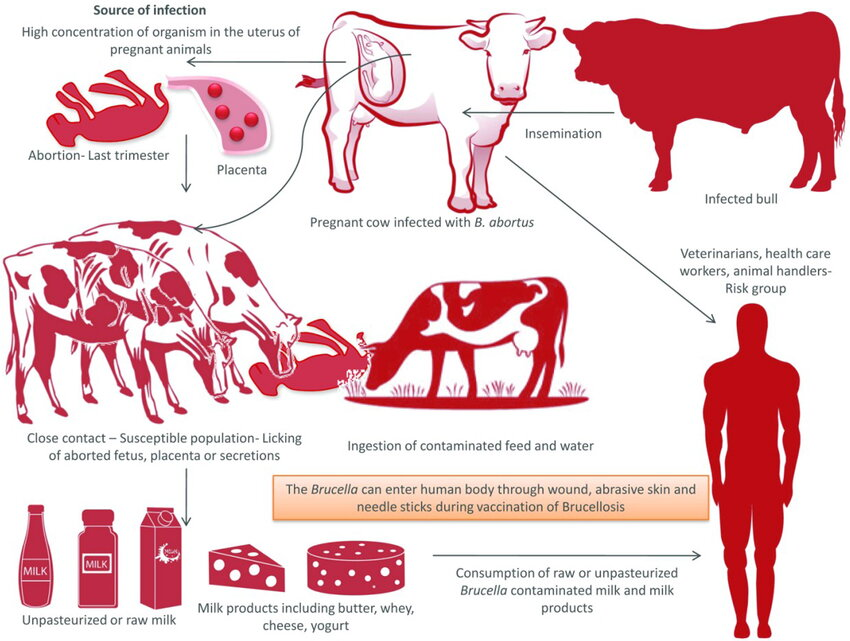
Figure: Transmission of Brucellosis: This diagram illustrates key routes of infection, including contact with aborted fetuses, contaminated milk products, and infected animals.
Despite ongoing control efforts, brucellosis remains a persistent zoonotic threat, highlighting the need for continued research and international collaboration to develop more effective prevention and control strategies.
Viral Hemorrhagic Fevers (VHFs): A Global Health Security Threat
Viral Hemorrhagic Fevers (VHFs), including Ebola and Rift Valley fever, are consistently prioritized due to their epidemic potential and the severity of illness they cause. These diseases are characterized by fever, bleeding disorders, and can progress to shock and death. The risk of VHFs is particularly pronounced in Africa, where outbreaks can have devastating consequences on public health systems and economies.
Key aspects of VHFs include:
- High Mortality: Many VHFs have high fatality rates, making them a significant public health concern.
- Rapid Spread: Some VHFs can spread rapidly through human-to-human transmission, particularly in healthcare settings.
- Limited Treatment Options: Effective treatments are often limited, emphasizing the importance of prevention and early detection.
Anthrax: A Zoonotic and Bioterrorism Threat
Anthrax, caused by the bacterium Bacillus anthracis, is another zoonotic disease of global concern. It primarily affects livestock but can also infect humans through contact with infected animals or contaminated products. Anthrax is also considered a potential bioterrorism agent due to its ability to form highly resistant spores that can be easily disseminated.
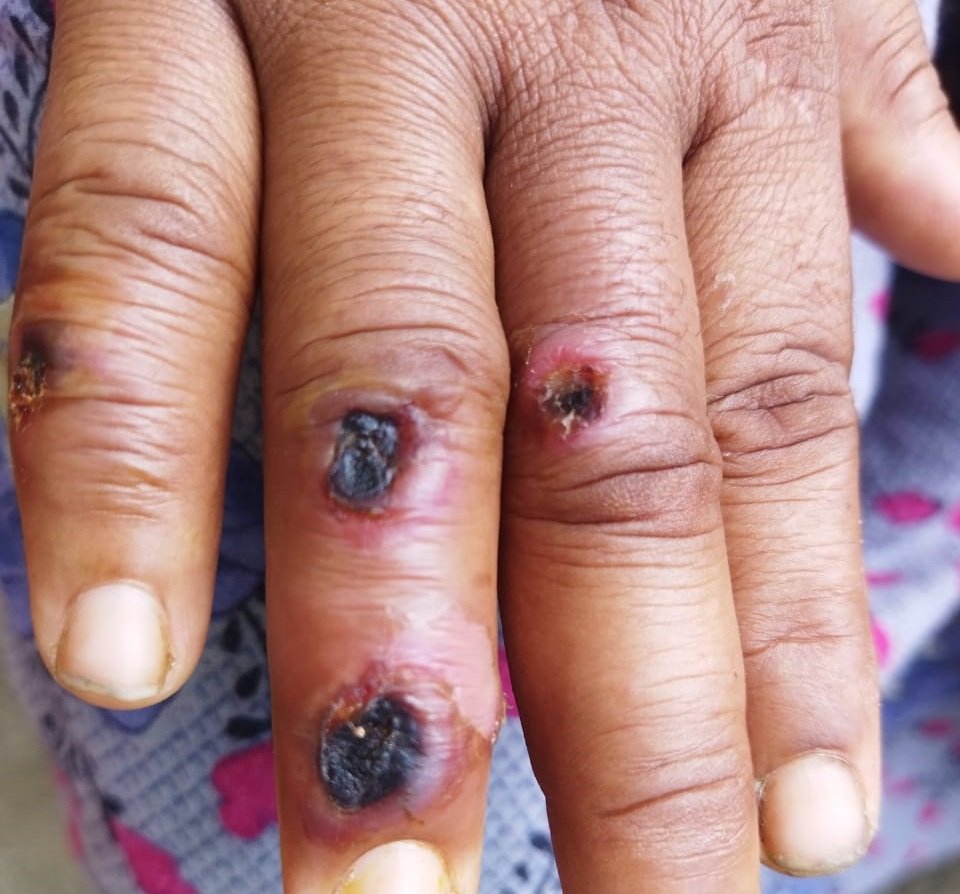
Key aspects of anthrax include:
- Environmental Persistence: Anthrax spores can survive in the environment for decades, posing a long-term risk.
- Multiple Forms of Infection: Anthrax can manifest in cutaneous, inhalation, and gastrointestinal forms, each with varying severity.
- Potential for Bioterrorism: The ease of dissemination and high mortality rate of inhalation anthrax make it a potential bioterrorism threat.
Risk Factors for Zoonotic Disease Transmission
We’ve explored how zoonotic diseases spread, but what makes some situations riskier than others? What factors increase the likelihood of these diseases jumping from animals to humans? Who are the individuals are at risk of having a zoonotic infection more likely than others? How does something like intensive farming practices increase the risk of zoonotic disease outbreaks? Let’s explore the factors that contribute to this increased risk.
As discussed in our article on emerging infectious diseases, several factors drive the spread of these illnesses. In addition to those factors, the CDC has identified specific at-risk groups.
- Children below 5 years
- Professionals closely working with animals like veterinarians, livestock farmers, wildlife rehabilitators, zookeepers etc
- Immunocompromised individual (e.g., HIV/AIDS, cancer treatment, organ transplantation)
- Pregnant women
- Adults over 65 years
The Impact of Zoonotic Diseases
Zoonotic diseases have far-reaching consequences for public health, agriculture, and global economies. Outbreaks of diseases such as rabies, avian influenza, and brucellosis place significant strain on healthcare systems and disrupt trade and food security. The economic toll is substantial, with losses in livestock productivity, increased healthcare costs, and market restrictions affecting national and global economies. In regions where zoonotic diseases are endemic, communities face long-term socio-economic burdens due to reduced labor capacity, disrupted food supply chains, and the necessity of expensive disease control measures. Moreover, emerging zoonotic threats such as Ebola and COVID-19 have demonstrated the potential for global pandemics, underscoring the urgency of preventive strategies
Prevention and Control Measures
Mitigating the risks associated with zoonotic diseases requires a comprehensive approach encompassing biosecurity, vaccination, surveillance, and education. Preventative measures begin at the individual level, with practices such as proper hand hygiene, safe food handling, and avoiding direct contact with sick animals. At the farm and industrial level, maintaining high biosecurity standards, implementing regular disease surveillance, and enforcing vaccination programs for livestock and pets are crucial in reducing the spillover of pathogens.
Public health interventions must also focus on reducing human exposure to zoonotic diseases through improved sanitation, vector control strategies, and community awareness campaigns. Additionally, regulatory policies should be strengthened to ensure safe wildlife trade and sustainable agricultural practices that limit human-wildlife interactions, which are a key driver of emerging zoonotic diseases.
Way forward through One Health Approach
Given the interconnected nature of human, animal, and environmental health, a One Health approach is essential in addressing zoonotic diseases. This interdisciplinary strategy calls for collaboration between veterinarians, medical professionals, environmental scientists, and policymakers to develop holistic solutions. Strengthening disease surveillance systems that integrate human and animal health data will enhance early detection and response to emerging threats.
Advancing research on zoonotic pathogens and their transmission dynamics can guide the development of effective vaccines and treatments. Furthermore, promoting sustainable land use and reducing habitat destruction will help mitigate the risks of pathogen spillover from wildlife to humans. By fostering cross-sectoral partnerships at local, national, and international levels, the One Health approach provides a roadmap for tackling zoonotic diseases more efficiently and preventing future pandemics.
Call for action
Addressing zoonotic diseases requires urgent action from governments, researchers, healthcare professionals, and the public. Policymakers must prioritize investment in One Health initiatives, strengthen biosecurity measures, and enhance surveillance and rapid response mechanisms. Researchers should continue investigating zoonotic pathogens to develop innovative diagnostic tools and effective vaccines. Healthcare professionals play a vital role in raising awareness and ensuring early diagnosis and treatment of zoonotic infections. Meanwhile, individuals can contribute by practicing safe hygiene, adhering to public health recommendations, and supporting sustainable farming and conservation efforts. The global community must unite to mitigate the threat of zoonotic diseases, ensuring a healthier and more resilient future for both humans and animals.
Zoonotic diseases remain a major global health concern, requiring continuous monitoring, research, and intervention. Strengthening the One Health approach will be essential in mitigating future outbreaks, ensuring both public health and economic stability worldwide. By addressing the underlying drivers of zoonotic disease emergence, we can reduce spillover events and safeguard global health security.